On June 26, 2013, William became the first patient in Canada to have NanoKnife treatment. It took two hours and he was awake throughout the procedure. (Video: UHN)
When William Dyke and his wife Maureen moved to their farm in Grand Valley, ON, they were looking forward to escaping the growing city.
In 2013, William was diagnosed with inoperable liver cancer – the simple life they had planned was now put on hold.
"I can remember our drive home after learning Bill had cancer," says Maureen. "Neither of us said much. We were both stunned by the news."
Before being diagnosed with Stage 1 liver cancer, William, a retired veterinarian assistant, was referred to a specialist at Toronto General Hospital when his family doctor received blood test results. William had Hepatitis C.
Despite treatment and close monitoring, an abdominal CT during a check-up revealed the feared news: he had developed liver cancer.
That's when William met Dr. Robert Beecroft.
When gold isn't good enough
"Only 25 to 40 per cent of patients with liver cancer are candidates for surgery. Often tumour number, location, spread outside the liver, or the degree of underlying liver damage from chronic liver disease preclude patients from having surgical treatment for liver cancer " says Dr. Beecroft, Interventional Radiologist, Joint Department of Medical Imaging.
With surgery – the current gold standard – not possible, Dr. Beecroft turned to less invasive interventional techniques to treat William's cancer.
Dr. Beecroft initially used percutaneous ethanol injection, administering alcohol directly into the tumour to dehydrate the cancer and kill it. Unfortunately in William's case, this approach had limitations and the cancer recurred.
The tumour's location further prevented other minimally invasive approaches including radiofrequency ablation (RFA) and thermal ablation (killing the tumour cells with heat by placing a special needle into the tumour) from being considered. This technique was too risky due to the tumour's close proximity to healthy structures, such as central bile ducts.
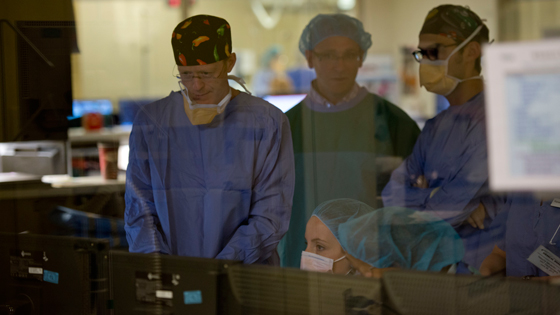
Dr. Beecroft and team review William’s CT scan during the NanoKnife procedure
Bring in the NanoKnife
As treatment options dwindled for William, Dr. Beecroft received some welcome news. He was approved for a pilot research trial using a treatment called the NanoKnife.
The NanoKnife is a form of non-thermal ablation that treats cancers located close to delicate structures – ideal for patients who aren't eligible for Radio Frequency Ablation (RFA) and surgery, like William's case.
The tool consists of two to four needles (19 gauge) inserted through the skin, each about two-centimetres apart to surround the tumour. High-voltage electrical impulses are delivered between the needles that kill the cancer cells without damaging surrounding structures.
The technique works by causing the opening of small pores that are nanometers in size (hence the name 'NanoKnife') in the cell membrane of the tumour cells, leading to cell death.
Dr. Beecroft asked if William would like to participate in the clinical trial. Without hesitation, William agreed.
"Maureen did a little bit of research about the NanoKnife on the internet," says William. "We knew there were cases being completed in the States, but it was Dr. Beecroft who gave us confidence in the treatment."
"He took the time to explain the procedure to us in detail, and go over its risks. He made both of us feel very comfortable," he says.
The trial was a Canadian-first and was set-up to determine NanoKnife's feasibility.
"We wanted to know: Can we actually perform this treatment? Is it effective? Can we perform it safely?" says Dr. Beecroft.

William Dyke, on his farm in Grand Valley, ON, is the first patient in Canada to have NanoKnife treatment. (Photo: William Dyke)
Cancer free after two hours
On June 26, 2013, William became the first patient in Canada to have NanoKnife treatment. It took two hours and he was awake throughout the procedure.
Before end of day, William was back on his farm in Grand Valley. The only medication prescribed for his treatment was Tylenol.
That was 18 months ago. William is now cancer free, and has check-ups with Dr. Beecroft every three months.
The 70-year- old retired veterinarian assistant and his wife are back on track with their plans for the quiet life.
"If it wasn't for Dr. Beecroft and the team, I'm not sure where I would be," says William. "I'm just so grateful for the attentiveness and care they showed me during the hardest part of my and my family's life."
Six liver cancer patients were treated in the trial, all completed as out-patients with same- day discharge, and with no reoccurrences of any of the treated tumours.
"The results of the trial indicate that we can perform this treatment, it's effective and it can be completed safely," says Dr. Beecroft. "For patients whose liver cancer is not able to be treated surgically or by thermal ablation due to location, we believe NanoKnife may be the best option."
Currently over 2,000 NanoKnife cases have been completed worldwide in the U.S., Europe, Australia, and now Canada.
Dr. Beecroft will present his clinical research results at the
World Congress of Interventional Oncology, May 6-9, in New York City.
